Journal Description
Viruses
Viruses
is a peer-reviewed, open access journal of virology, published monthly online by MDPI. The American Society for Virology (ASV), Spanish Society for Virology (SEV), Canadian Society for Virology (CSV), Italian Society for Virology (SIV-ISV), Australasian Virology Society (AVS) and others are affiliated with Viruses and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, MEDLINE, PMC, Embase, PubAg, AGRIS, and other databases.
- Journal Rank: JCR - Q2 (Virology) / CiteScore - Q1 (Infectious Diseases)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 13.8 days after submission; acceptance to publication is undertaken in 2.5 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Companion journals for Viruses include: COVID and Zoonotic Diseases.
Impact Factor:
4.7 (2022);
5-Year Impact Factor:
4.8 (2022)
Latest Articles
Post-Transcriptional HIV-1 Latency: A Promising Target for Therapy?
Viruses 2024, 16(5), 666; https://doi.org/10.3390/v16050666 (registering DOI) - 24 Apr 2024
Abstract
Human Immunodeficiency Virus type 1 (HIV-1) latency represents a significant hurdle in finding a cure for HIV-1 infections, despite tireless research efforts. This challenge is partly attributed to the intricate nature of HIV-1 latency, wherein various host and viral factors participate in multiple
[...] Read more.
Human Immunodeficiency Virus type 1 (HIV-1) latency represents a significant hurdle in finding a cure for HIV-1 infections, despite tireless research efforts. This challenge is partly attributed to the intricate nature of HIV-1 latency, wherein various host and viral factors participate in multiple physiological processes. While substantial progress has been made in discovering therapeutic targets for HIV-1 transcription, targets for the post-transcriptional regulation of HIV-1 infections have received less attention. However, cumulative evidence now suggests the pivotal contribution of post-transcriptional regulation to the viral latency in both in vitro models and infected individuals. In this review, we explore recent insights on post-transcriptional latency in HIV-1 and discuss the potential of its therapeutic targets, illustrating some host factors that restrict HIV-1 at the post-transcriptional level.
Full article
(This article belongs to the Special Issue Unraveling the Pathogenesis of Persistent Virus Infection)
Open AccessArticle
Exploring the Antiviral Potential of Esters of Cinnamic Acids with Quercetin
by
Valeria Manca, Annalisa Chianese, Vanessa Palmas, Federica Etzi, Carla Zannella, Davide Moi, Francesco Secci, Gabriele Serreli, Giorgia Sarais, Maria Vittoria Morone, Massimiliano Galdiero, Valentina Onnis, Aldo Manzin and Giuseppina Sanna
Viruses 2024, 16(5), 665; https://doi.org/10.3390/v16050665 - 24 Apr 2024
Abstract
Severe acute respiratory syndrome-related Coronavirus 2 (SARS-CoV-2) has infected more than 762 million people to date and has caused approximately 7 million deaths all around the world, involving more than 187 countries. Although currently available vaccines show high efficacy in preventing severe respiratory
[...] Read more.
Severe acute respiratory syndrome-related Coronavirus 2 (SARS-CoV-2) has infected more than 762 million people to date and has caused approximately 7 million deaths all around the world, involving more than 187 countries. Although currently available vaccines show high efficacy in preventing severe respiratory complications in infected patients, the high number of mutations in the S proteins of the current variants is responsible for the high level of immune evasion and transmissibility of the virus and the reduced effectiveness of acquired immunity. In this scenario, the development of safe and effective drugs of synthetic or natural origin to suppress viral replication and treat acute forms of COVID-19 remains a valid therapeutic challenge. Given the successful history of flavonoids-based drug discovery, we developed esters of substituted cinnamic acids with quercetin to evaluate their in vitro activity against a broad spectrum of Coronaviruses. Interestingly, two derivatives, the 3,4-methylenedioxy 6 and the ester of acid 7, have proved to be effective in reducing OC43-induced cytopathogenicity, showing interesting EC50s profiles. The ester of synaptic acid 7 in particular, which is not endowed with relevant cytotoxicity under any of the tested conditions, turned out to be active against OC43 and SARS-CoV-2, showing a promising EC50. Therefore, said compound was selected as the lead object of further analysis. When tested in a yield reduction, assay 7 produced a significant dose-dependent reduction in viral titer. However, the compound was not virucidal, as exposure to high concentrations of it did not affect viral infectivity, nor did it affect hCoV-OC43 penetration into pre-treated host cells. Additional studies on the action mechanism have suggested that our derivative may inhibit viral endocytosis by reducing viral attachment to host cells.
Full article
(This article belongs to the Special Issue Recent Advances in Antiviral Natural Products 2023)
Open AccessArticle
Dynamics of Matricellular Protein Levels in Blood Predict Recovery in Patients with Human Immunodeficiency Virus-Tuberculosis Coinfection
by
Ashwini Shete, Manisha Ghate, Hiroko Iwasaki-Hozumi, Sandip Patil, Pallavi Shidhaye, Gaowa Bai, Takashi Matsuba, Pratiksha Pharande, Bharati Mahajan, Aarti Randive, Anupam Mukherjee and Toshio Hattori
Viruses 2024, 16(5), 664; https://doi.org/10.3390/v16050664 - 24 Apr 2024
Abstract
Chronic immune activation in tuberculosis (TB) associated with human immunodeficiency virus (HIV) infection (HIV/TB) modifies their clinical course. We prospectively measured osteopontin (OPN), full-length galectin-9 (FL-Gal9), and total-Gal9 (T-Gal9) levels in 32 patients with HIV/TB coinfection treated with anti-tuberculosis and antiretroviral therapies over
[...] Read more.
Chronic immune activation in tuberculosis (TB) associated with human immunodeficiency virus (HIV) infection (HIV/TB) modifies their clinical course. We prospectively measured osteopontin (OPN), full-length galectin-9 (FL-Gal9), and total-Gal9 (T-Gal9) levels in 32 patients with HIV/TB coinfection treated with anti-tuberculosis and antiretroviral therapies over 6–18 months to determine the amelioration of inflammatory conditions in response to the therapies. We observed a significant time-dependent decrease in FL-Gal9 in both pulmonary TB (PTB, n = 20) and extrapulmonary TB (EPTB, n = 12) patients. The levels of T-Gal9, OPN, and CRP decreased significantly after treatment in only PTB patients. We calculated the inflammatory score (INS) indicating immunologic recovery based on the decline in OPN, FL-Gal9, T-Gal9, and CRP levels. Baseline levels of T-Gal9 and OPN positively correlated with INS in all TB and only PTB patients, respectively, indicating that their levels predict better recovery. In contrast, FL-Gal9 levels at the second visit negatively correlated with INS in EPTB patients. The decrease rate in OPN levels at the second visit also correlated positively with INS in PTB patients. Women showed a higher INS and lower levels of FL-Gal9 than men. The patients with moderate grade severity on chest X-ray had higher CD4 cell numbers than those with limited grade severity. Monitoring these markers will help to predict and assess the response to therapy as well as to devise strategies to reduce the complications caused by chronic immune activation in patients with HIV/TB coinfection.
Full article
(This article belongs to the Special Issue Tuberculosis (TB) and HIV Coinfection)
Open AccessReview
Mechanisms by Which SARS-CoV-2 Invades and Damages the Central Nervous System: Apart from the Immune Response and Inflammatory Storm, What Else Do We Know?
by
Zihan Sun, Chunying Shi and Lixin Jin
Viruses 2024, 16(5), 663; https://doi.org/10.3390/v16050663 - 24 Apr 2024
Abstract
Initially reported as pneumonia of unknown origin, COVID-19 is increasingly being recognized for its impact on the nervous system, despite nervous system invasions being extremely rare. As a result, numerous studies have been conducted to elucidate the mechanisms of nervous system damage and
[...] Read more.
Initially reported as pneumonia of unknown origin, COVID-19 is increasingly being recognized for its impact on the nervous system, despite nervous system invasions being extremely rare. As a result, numerous studies have been conducted to elucidate the mechanisms of nervous system damage and propose appropriate coping strategies. This review summarizes the mechanisms by which SARS-CoV-2 invades and damages the central nervous system, with a specific focus on aspects apart from the immune response and inflammatory storm. The latest research findings on these mechanisms are presented, providing new insights for further in-depth research.
Full article
(This article belongs to the Section Coronaviruses)
Open AccessArticle
Design and Development of an Antigen Test for SARS-CoV-2 Nucleocapsid Protein to Validate the Viral Quality Assurance Panels
by
Partha Ray, Melissa Ledgerwood-Lee, Howard Brickner, Alex E. Clark, Aaron Garretson, Rishi Graham, Westley Van Zant, Aaron F. Carlin and Eliah S. Aronoff-Spencer
Viruses 2024, 16(5), 662; https://doi.org/10.3390/v16050662 - 24 Apr 2024
Abstract
The continuing mutability of the SARS-CoV-2 virus can result in failures of diagnostic assays. To address this, we describe a generalizable bioinformatics-to-biology pipeline developed for the calibration and quality assurance of inactivated SARS-CoV-2 variant panels provided to Radical Acceleration of Diagnostics programs (RADx)-radical
[...] Read more.
The continuing mutability of the SARS-CoV-2 virus can result in failures of diagnostic assays. To address this, we describe a generalizable bioinformatics-to-biology pipeline developed for the calibration and quality assurance of inactivated SARS-CoV-2 variant panels provided to Radical Acceleration of Diagnostics programs (RADx)-radical program awardees. A heuristic genetic analysis based on variant-defining mutations demonstrated the lowest genetic variance in the Nucleocapsid protein (Np)- C-terminal domain (CTD) across all SARS-CoV-2 variants. We then employed the Shannon entropy method on (Np) sequences collected from the major variants, verifying the CTD with lower entropy (less prone to mutations) than other Np regions. Polyclonal and monoclonal antibodies were raised against this target CTD antigen and used to develop an Enzyme-linked immunoassay (ELISA) test for SARS-CoV-2. Blinded Viral Quality Assurance (VQA) panels comprised of UV-inactivated SARS-CoV-2 variants (XBB.1.5, BF.7, BA.1, B.1.617.2, and WA1) and distractor respiratory viruses (CoV 229E, CoV OC43, RSV A2, RSV B, IAV H1N1, and IBV) were assembled by the RADx-rad Diagnostics core and tested using the ELISA described here. The assay tested positive for all variants with high sensitivity (limit of detection: 1.72–8.78 ng/mL) and negative for the distractor virus panel. Epitope mapping for the monoclonal antibodies identified a 20 amino acid antigenic peptide on the Np-CTD that an in-silico program also predicted for the highest antigenicity. This work provides a template for a bioinformatics pipeline to select genetic regions with a low propensity for mutation (low Shannon entropy) to develop robust ‘pan-variant’ antigen-based assays for viruses prone to high mutational rates.
Full article
(This article belongs to the Special Issue Antibody-Based Therapeutics and Diagnostics for Viral Diseases)
►▼
Show Figures

Graphical abstract
Open AccessArticle
T-Cell Responses to COVID-19 Vaccines and Breakthrough Infection in People Living with HIV Receiving Antiretroviral Therapy
by
Sneha Datwani, Rebecca Kalikawe, Rachel Waterworth, Francis M. Mwimanzi, Richard Liang, Yurou Sang, Hope R. Lapointe, Peter K. Cheung, Fredrick Harrison Omondi, Maggie C. Duncan, Evan Barad, Sarah Speckmaier, Nadia Moran-Garcia, Mari L. DeMarco, Malcolm Hedgcock, Cecilia T. Costiniuk, Mark Hull, Marianne Harris, Marc G. Romney, Julio S. G. Montaner, Zabrina L. Brumme and Mark A. Brockmanadd
Show full author list
remove
Hide full author list
Viruses 2024, 16(5), 661; https://doi.org/10.3390/v16050661 - 24 Apr 2024
Abstract
People living with HIV (PLWH) can exhibit impaired immune responses to vaccines. Accumulating evidence indicates that PLWH, particularly those receiving antiretroviral therapy, mount strong antibody responses to COVID-19 vaccines, but fewer studies have examined cellular immune responses to the vaccinations. Here, we used
[...] Read more.
People living with HIV (PLWH) can exhibit impaired immune responses to vaccines. Accumulating evidence indicates that PLWH, particularly those receiving antiretroviral therapy, mount strong antibody responses to COVID-19 vaccines, but fewer studies have examined cellular immune responses to the vaccinations. Here, we used an activation-induced marker (AIM) assay to quantify SARS-CoV-2 spike-specific CD4+ and CD8+ T cells generated by two and three doses of COVID-19 vaccines in 50 PLWH receiving antiretroviral therapy, compared to 87 control participants without HIV. In a subset of PLWH, T-cell responses were also assessed after post-vaccine breakthrough infections and/or receipt of a fourth vaccine dose. All participants remained SARS-CoV-2 infection-naive until at least one month after their third vaccine dose. SARS-CoV-2 infection was determined by seroconversion to a Nucleocapsid (N) antigen, which occurred in 21 PLWH and 38 control participants after the third vaccine dose. Multivariable regression analyses were used to investigate the relationships between sociodemographic, health- and vaccine-related variables, vaccine-induced T-cell responses, and breakthrough infection risk. We observed that a third vaccine dose boosted spike-specific CD4+ and CD8+ T-cell frequencies significantly above those measured after the second dose (all p < 0.0001). Median T-cell frequencies did not differ between PLWH and controls after the second dose (p > 0.1), but CD8+ T-cell responses were modestly lower in PLWH after the third dose (p = 0.02), an observation that remained significant after adjusting for sociodemographic, health- and vaccine-related variables (p = 0.045). In PLWH who experienced a breakthrough infection, median T-cell frequencies increased even higher than those observed after three vaccine doses (p < 0.03), and CD8+ T-cell responses in this group remained higher even after a fourth vaccine dose (p = 0.03). In multivariable analyses, the only factor associated with an increased breakthrough infection risk was younger age, which is consistent with the rapid increase in SARS-CoV-2 seropositivity that was seen among younger adults in Canada after the initial appearance of the Omicron variant. These results indicate that PLWH receiving antiretroviral therapy mount strong T-cell responses to COVID-19 vaccines that can be enhanced by booster doses or breakthrough infection.
Full article
(This article belongs to the Special Issue Immunogenicity and Safety of COVID-19 Vaccine: A Milestone Achieved in the Battle against the Pandemic)
Open AccessCommunication
Histo-Blood Group Antigen-Producing Bacterial Cocktail Reduces Rotavirus A, B, and C Infection and Disease in Gnotobiotic Piglets
by
Sergei A. Raev, Maryssa K. Kick, Maria Chellis, Joshua O. Amimo, Linda J. Saif and Anastasia N. Vlasova
Viruses 2024, 16(5), 660; https://doi.org/10.3390/v16050660 - 24 Apr 2024
Abstract
The suboptimal performance of rotavirus (RV) vaccines in developing countries and in animals necessitates further research on the development of novel therapeutics and control strategies. To initiate infection, RV interacts with cell-surface O-glycans, including histo-blood group antigens (HBGAs). We have previously demonstrated
[...] Read more.
The suboptimal performance of rotavirus (RV) vaccines in developing countries and in animals necessitates further research on the development of novel therapeutics and control strategies. To initiate infection, RV interacts with cell-surface O-glycans, including histo-blood group antigens (HBGAs). We have previously demonstrated that certain non-pathogenic bacteria express HBGA- like substances (HBGA+) capable of binding RV particles in vitro. We hypothesized that HBGA+ bacteria can bind RV particles in the gut lumen protecting against RV species A (RVA), B (RVB), and C (RVC) infection in vivo. In this study, germ-free piglets were colonized with HBGA+ or HBGA- bacterial cocktail and infected with RVA/RVB/RVC of different genotypes. Diarrhea severity, virus shedding, immunoglobulin A (IgA) Ab titers, and cytokine levels were evaluated. Overall, colonization with HBGA+ bacteria resulted in reduced diarrhea severity and virus shedding compared to the HBGA- bacteria. Consistent with our hypothesis, the reduced severity of RV disease and infection was not associated with significant alterations in immune responses. Additionally, colonization with HBGA+ bacteria conferred beneficial effects irrespective of the piglet HBGA phenotype. These findings are the first experimental evidence that probiotic performance in vivo can be improved by including HBGA+ bacteria, providing decoy epitopes for broader/more consistent protection against diverse RVs.
Full article
(This article belongs to the Special Issue Rotaviruses and Rotavirus Vaccines)
►▼
Show Figures
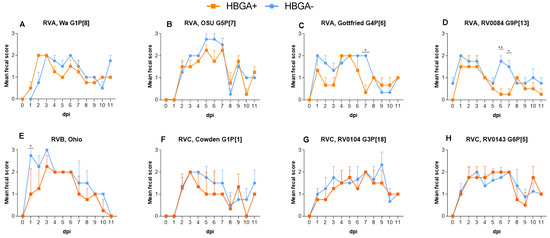
Figure 1
Open AccessReview
Design and Application of Biosafe Coronavirus Engineering Systems without Virulence
by
Guoqiang Wu, Qiaoyu Li, Junbiao Dai, Guobin Mao and Yingxin Ma
Viruses 2024, 16(5), 659; https://doi.org/10.3390/v16050659 - 24 Apr 2024
Abstract
In the last twenty years, three deadly zoonotic coronaviruses (CoVs)—namely, severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV), and SARS-CoV-2—have emerged. They are considered highly pathogenic for humans, particularly SARS-CoV-2, which caused the 2019 CoV disease pandemic (COVID-19), endangering
[...] Read more.
In the last twenty years, three deadly zoonotic coronaviruses (CoVs)—namely, severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV), and SARS-CoV-2—have emerged. They are considered highly pathogenic for humans, particularly SARS-CoV-2, which caused the 2019 CoV disease pandemic (COVID-19), endangering the lives and health of people globally and causing unpredictable economic losses. Experiments on wild-type viruses require biosafety level 3 or 4 laboratories (BSL-3 or BSL-4), which significantly hinders basic virological research. Therefore, the development of various biosafe CoV systems without virulence is urgently needed to meet the requirements of different research fields, such as antiviral and vaccine evaluation. This review aimed to comprehensively summarize the biosafety of CoV engineering systems. These systems combine virological foundations with synthetic genomics techniques, enabling the development of efficient tools for attenuated or non-virulent vaccines, the screening of antiviral drugs, and the investigation of the pathogenic mechanisms of novel microorganisms.
Full article
(This article belongs to the Special Issue Broad-Spectrum Antivirals and Interaction with Viruses)
►▼
Show Figures
Figure 1
Open AccessArticle
Expanding the Scope of Adenoviral Vectors by Utilizing Novel Tools for Recombination and Vector Rescue
by
Julian Fischer, Ariana Fedotova, Clara Bühler, Laura Darriba, Sabrina Schreiner and Zsolt Ruzsics
Viruses 2024, 16(5), 658; https://doi.org/10.3390/v16050658 - 23 Apr 2024
Abstract
Recombinant adenoviruses are widely used in clinical and laboratory applications. Despite the wide variety of available sero- and genotypes, only a fraction is utilized in vivo. As adenoviruses are a large group of viruses, displaying many different tropisms, immune epitopes, and replication characteristics,
[...] Read more.
Recombinant adenoviruses are widely used in clinical and laboratory applications. Despite the wide variety of available sero- and genotypes, only a fraction is utilized in vivo. As adenoviruses are a large group of viruses, displaying many different tropisms, immune epitopes, and replication characteristics, the merits of translating these natural benefits into vector applications are apparent. This translation, however, proves difficult, since while research has investigated the application of these viruses, there are no universally applicable rules in vector design for non-classical adenovirus types. In this paper, we describe a generalized workflow that allows vectorization, rescue, and cloning of all adenoviral species to enable the rapid development of new vector variants. We show this using human and simian adenoviruses, further modifying a selection of them to investigate their gene transfer potential and build potential vector candidates for future applications.
Full article
(This article belongs to the Special Issue 15th International Adenovirus Meeting)
Open AccessArticle
Wide Real-Life Data Support Reduced Sensitivity of Antigen Tests for Omicron SARS-CoV-2 Infections
by
Chiara Piubelli, Davide Treggiari, Denise Lavezzari, Michela Deiana, Klevia Dishnica, Emma Maria Sole Tosato, Cristina Mazzi, Paolo Cattaneo, Antonio Mori, Elena Pomari, Lavinia Nicolini, Martina Leonardi, Francesca Perandin, Fabio Formenti, Alejandro Giorgetti, Antonio Conti, Maria Rosaria Capobianchi, Federico Giovanni Gobbi and Concetta Castilletti
Viruses 2024, 16(5), 657; https://doi.org/10.3390/v16050657 - 23 Apr 2024
Abstract
With the continuous spread of new SARS-CoV-2 variants of concern (VOCs), the monitoring of diagnostic test performances is mandatory. We evaluated the changes in antigen diagnostic tests’ (ADTs) accuracy along the Delta to Omicron VOCs transition, exploring the N protein mutations possibly affecting
[...] Read more.
With the continuous spread of new SARS-CoV-2 variants of concern (VOCs), the monitoring of diagnostic test performances is mandatory. We evaluated the changes in antigen diagnostic tests’ (ADTs) accuracy along the Delta to Omicron VOCs transition, exploring the N protein mutations possibly affecting ADT sensitivity and assessing the best sampling site for the diagnosis of Omicron infections. In total, 5175 subjects were enrolled from 1 October 2021 to 15 July 2022. The inclusion criteria were SARS-CoV-2 ADT combined with a same-day RT-PCR swab test. For the sampling site analysis, 61 patients were prospectively recruited during the Omicron period for nasal and oral swab analyses by RT-PCR. Next-Generation Sequencing data were obtained to evaluate the different sublineages. Using RT-PCR as a reference, 387 subjects resulted in becoming infected and the overall sensitivity of the ADT decreased from 63% in the Delta period to 33% in the Omicron period. This decrease was highly statistically significant (p < 0.001), and no decrease in viral load was detected at the RNA level. The nasal site presented a significantly higher viral load than the oral site during the Omicron wave. The reduced detection rate of Omicron infections by ADT should be considered in the global testing strategy to preserve accurate diagnoses across the changing SARS-CoV-2 variants.
Full article
(This article belongs to the Section Coronaviruses)
►▼
Show Figures
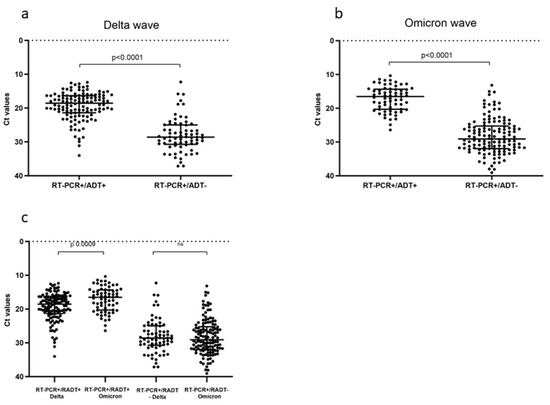
Figure 1
Open AccessArticle
The Changing Detection Rate of Respiratory Syncytial Virus in Adults in Western Australia between 2017 and 2023
by
David A. Foley, Cara A. Minney-Smith, Andrew Tjea, Mark P. Nicol, Avram Levy, Hannah C. Moore and Christopher C. Blyth
Viruses 2024, 16(5), 656; https://doi.org/10.3390/v16050656 - 23 Apr 2024
Abstract
The incidence of respiratory syncytial virus (RSV) in adults is inadequately defined and the impact of SARS-CoV-2-related non-pharmaceutical interventions (NPIs) is underexplored. Using laboratory data, we described the detection rate of RSV in adults ≥16 years in Western Australia (WA) between 2017 and
[...] Read more.
The incidence of respiratory syncytial virus (RSV) in adults is inadequately defined and the impact of SARS-CoV-2-related non-pharmaceutical interventions (NPIs) is underexplored. Using laboratory data, we described the detection rate of RSV in adults ≥16 years in Western Australia (WA) between 2017 and 2023. With the exception of 2020, RSV detections rose annually between 2017 and 2023, reaching 50.7 per 100,000 in 2023 (95% confidence interval [CI], 47.9–53.8). RSV testing expanded considerably across the study period, with the testing in 2023 more than five times the 2017 total. The detection rate was highest in adults ≥60 years between 2017 and 2019, particularly those ≥75 years. Following 2020, the detections in all age groups increased, with the highest detection rate in 2023 in those ≥75-years (199.5 per 100,000; 95% CI, 180.5–220). NPIs significantly impacted RSV seasonality; the preceding winter pattern was disrupted, resulting in an absent 2020 winter season and two major summer seasons in 2020/21 and 2021/22. The RSV season began to realign in 2022, reverting to a winter seasonal pattern in 2023 and the largest season in the study period. Ongoing surveillance will be required to understand the stability of these increases and to delineate the impact of new immunisation strategies.
Full article
(This article belongs to the Special Issue Impact of Pandemic Measures on the Epidemiology and Seasonality of Other Viruses)
►▼
Show Figures
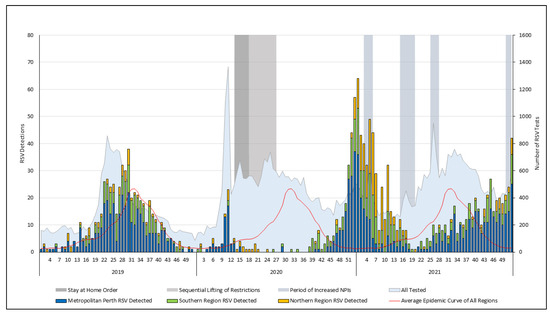
Figure 1
Open AccessArticle
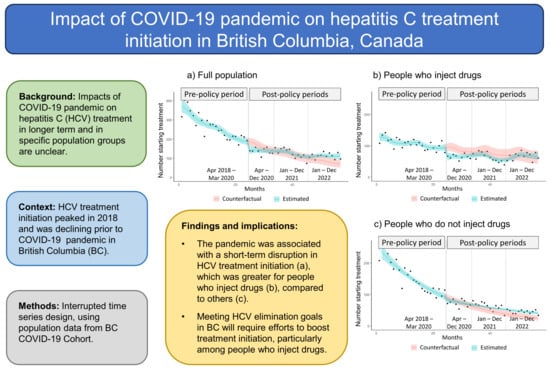
Impact of the COVID-19 Pandemic on Hepatitis C Treatment Initiation in British Columbia, Canada: An Interrupted Time Series Study
by
Richard L. Morrow, Mawuena Binka, Julia Li, Mike Irvine, Sofia R. Bartlett, Stanley Wong, Dahn Jeong, Jean Damascene Makuza, Jason Wong, Amanda Yu, Mel Krajden and Naveed Zafar Janjua
Viruses 2024, 16(5), 655; https://doi.org/10.3390/v16050655 - 23 Apr 2024
Abstract
We investigated the impacts of the COVID-19 pandemic on hepatitis C (HCV) treatment initiation, including by birth cohort and injection drug use status, in British Columbia (BC), Canada. Using population data from the BC COVID-19 Cohort, we conducted interrupted time series analyses, estimating
[...] Read more.
We investigated the impacts of the COVID-19 pandemic on hepatitis C (HCV) treatment initiation, including by birth cohort and injection drug use status, in British Columbia (BC), Canada. Using population data from the BC COVID-19 Cohort, we conducted interrupted time series analyses, estimating changes in HCV treatment initiation following the introduction of pandemic-related policies in March 2020. The study included a pre-policy period (April 2018 to March 2020) and three follow-up periods (April to December 2020, January to December 2021, and January to December 2022). The level of HCV treatment initiation decreased by 26% in April 2020 (rate ratio 0.74, 95% confidence interval [CI] 0.60 to 0.91). Overall, no statistically significant difference in HCV treatment initiation occurred over the 2020 and 2021 post-policy periods, and an increase of 34.4% (95% CI 0.6 to 75.8) occurred in 2022 (equating to 321 additional people initiating treatment), relative to expectation. Decreases in HCV treatment initiation occurred in 2020 for people born between 1965 and 1974 (25.5%) and people who inject drugs (24.5%), relative to expectation. In summary, the pandemic was associated with short-term disruptions in HCV treatment initiation in BC, which were greater for people born 1965 to 1974 and people who inject drugs.
Full article
(This article belongs to the Special Issue Cascade of Care for HIV and Hepatitis)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Epidemiological Features of Human Norovirus Genotypes before and after COVID-19 Countermeasures in Osaka, Japan
by
Tatsuya Shirai, Juthamas Phadungsombat, Yumi Ushikai, Kunihito Yoshikaie, Tatsuo Shioda and Naomi Sakon
Viruses 2024, 16(4), 654; https://doi.org/10.3390/v16040654 - 22 Apr 2024
Abstract
We investigated the molecular epidemiology of human norovirus (HuNoV) in all age groups using samples from April 2019 to March 2023, before and after the COVID-19 countermeasures were implemented. GII.2[P16] and GII.4[P31], the prevalent strains in Japan before COVID-19 countermeasures, remained prevalent during
[...] Read more.
We investigated the molecular epidemiology of human norovirus (HuNoV) in all age groups using samples from April 2019 to March 2023, before and after the COVID-19 countermeasures were implemented. GII.2[P16] and GII.4[P31], the prevalent strains in Japan before COVID-19 countermeasures, remained prevalent during the COVID-19 pandemic, except from April to November 2020; in 2021, the prevalence of GII.2[P16] increased among children. Furthermore, there was an increase in the prevalence of GII.4[P16] after December 2022. Phylogenetic analysis of GII.P31 RdRp showed that some strains detected in 2022 belonged to a different cluster of other strains obtained during the present study period, suggesting that HuNoV strains will evolve differently even if they have the same type of RdRp. An analysis of the amino acid sequence of VP1 showed that some antigenic sites of GII.4[P16] were different from those of GII.4[P31]. The present study showed high infectivity of HuNoV despite the COVID-19 countermeasures and revealed changes in the prevalent genotypes and mutations of each genotype. In the future, we will investigate whether GII.4[P16] becomes more prevalent, providing new insights by comparing the new data with those analyzed in the present study.
Full article
(This article belongs to the Special Issue Viral Genetic Variation)
►▼
Show Figures

Figure 1
Open AccessArticle
Delving into the Aftermath of a Disease-Associated Near-Extinction Event: A Five-Year Study of a Serpentovirus (Nidovirus) in a Critically Endangered Turtle Population
by
Kate Parrish, Peter Kirkland, Paul Horwood, Bruce Chessman, Shane Ruming, Gerry McGilvray, Karrie Rose, Jane Hall and Lee Skerratt
Viruses 2024, 16(4), 653; https://doi.org/10.3390/v16040653 - 22 Apr 2024
Abstract
Bellinger River virus (BRV) is a serpentovirus (nidovirus) that was likely responsible for the catastrophic mortality of the Australian freshwater turtle Myuchelys georgesi in February 2015. From November 2015 to November 2020, swabs were collected from turtles during repeated river surveys to estimate
[...] Read more.
Bellinger River virus (BRV) is a serpentovirus (nidovirus) that was likely responsible for the catastrophic mortality of the Australian freshwater turtle Myuchelys georgesi in February 2015. From November 2015 to November 2020, swabs were collected from turtles during repeated river surveys to estimate the prevalence of BRV RNA, identify risk factors associated with BRV infection, and refine sample collection. BRV RNA prevalence at first capture was significantly higher in M. georgesi (10.8%) than in a coexisting turtle, Emydura macquarii (1.0%). For M. georgesi, various risk factors were identified depending on the analysis method, but a positive BRV result was consistently associated with a larger body size. All turtles were asymptomatic when sampled and conjunctival swabs were inferred to be optimal for ongoing monitoring. Although the absence of disease and recent BRV detections suggests a reduced ongoing threat, the potential for the virus to persist in an endemic focus or resurge in cyclical epidemics cannot be excluded. Therefore, BRV is an ongoing potential threat to the conservation of M. georgesi, and strict adherence to biosecurity principles is essential to minimise the risk of reintroduction or spread of BRV or other pathogens.
Full article
(This article belongs to the Special Issue Identifying and Characterizing Viral Infections in Reptiles, Amphibians, Fish and Other Aquatic Species)
►▼
Show Figures
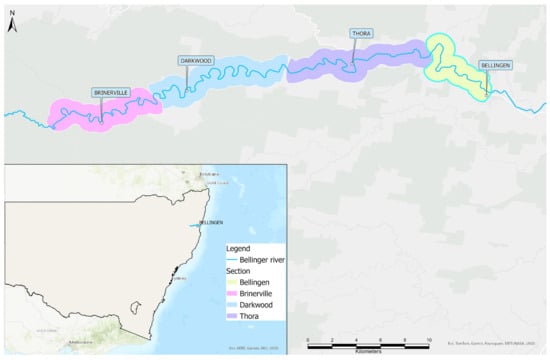
Figure 1
Open AccessArticle
Heterologous Exchanges of Glycoprotein and Non-Virion Protein in Novirhabdoviruses: Assessment of Virulence in Yellow Perch (Perca flavescens) and Rainbow Trout (Oncorhynchus mykiss)
by
Vikram N. Vakharia, Arun Ammayappan, Shamila Yusuff, Tarin M. Tesfaye and Gael Kurath
Viruses 2024, 16(4), 652; https://doi.org/10.3390/v16040652 - 22 Apr 2024
Abstract
Infectious hematopoietic necrosis virus (IHNV) and viral hemorrhagic septicemia virus (VHSV) are rhabdoviruses in two different species belonging to the Novirhabdovirus genus. IHNV has a narrow host range restricted to trout and salmon species, and viruses in the M genogroup of IHNV have
[...] Read more.
Infectious hematopoietic necrosis virus (IHNV) and viral hemorrhagic septicemia virus (VHSV) are rhabdoviruses in two different species belonging to the Novirhabdovirus genus. IHNV has a narrow host range restricted to trout and salmon species, and viruses in the M genogroup of IHNV have high virulence in rainbow trout (Oncorhynchus mykiss). In contrast, the VHSV genotype IVb that invaded the Great Lakes in the United States has a broad host range, with high virulence in yellow perch (Perca flavescens), but not in rainbow trout. By using reverse-genetic systems of IHNV-M and VHSV-IVb strains, we generated six IHNV:VHSV chimeric viruses in which the glycoprotein (G), non-virion-protein (NV), or both G and NV genes of IHNV-M were replaced with the analogous genes from VHSV-IVb, and vice versa. These chimeric viruses were used to challenge groups of rainbow trout and yellow perch. The parental recombinants rIHNV-M and rVHSV-IVb were highly virulent in rainbow trout and yellow perch, respectively. Parental rIHNV-M was avirulent in yellow perch, and chimeric rIHNV carrying G, NV, or G and NV genes from VHSV-IVb remained low in virulence in yellow perch. Similarly, the parental rVHSV-IVb exhibited low virulence in rainbow trout, and chimeric rVHSV with substituted G, NV, or G and NV genes from IHNV-M remained avirulent in rainbow trout. Thus, the G and NV genes of either virus were not sufficient to confer high host-specific virulence when exchanged into a heterologous species genome. Some exchanges of G and/or NV genes caused a loss of host-specific virulence, providing insights into possible roles in viral virulence or fitness, and interactions between viral proteins.
Full article
(This article belongs to the Special Issue The World of Rhabdoviruses)
►▼
Show Figures
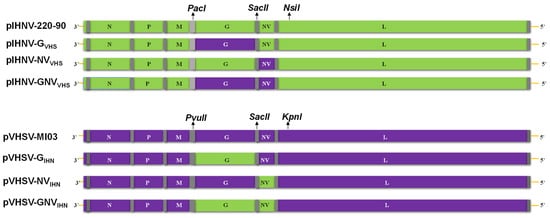
Figure 1
Open AccessArticle
Why Certain Repurposed Drugs Are Unlikely to Be Effective Antivirals to Treat SARS-CoV-2 Infections
by
Selwyn J. Hurwitz, Ramyani De, Julia C. LeCher, Jessica A. Downs-Bowen, Shu Ling Goh, Keivan Zandi, Tamara McBrayer, Franck Amblard, Dharmeshkumar Patel, James J. Kohler, Manoj Bhasin, Brian S. Dobosh, Vikas Sukhatme, Rabindra M. Tirouvanziam and Raymond F. Schinazi
Viruses 2024, 16(4), 651; https://doi.org/10.3390/v16040651 - 22 Apr 2024
Abstract
Most repurposed drugs have proved ineffective for treating COVID-19. We evaluated median effective and toxic concentrations (EC50, CC50) of 49 drugs, mostly from previous clinical trials, in Vero cells. Ratios of reported unbound peak plasma concentrations, (Cmax)/EC
[...] Read more.
Most repurposed drugs have proved ineffective for treating COVID-19. We evaluated median effective and toxic concentrations (EC50, CC50) of 49 drugs, mostly from previous clinical trials, in Vero cells. Ratios of reported unbound peak plasma concentrations, (Cmax)/EC50, were used to predict the potential in vivo efficacy. The 20 drugs with the highest ratios were retested in human Calu-3 and Caco-2 cells, and their CC50 was determined in an expanded panel of cell lines. Many of the 20 drugs with the highest ratios were inactive in human Calu-3 and Caco-2 cells. Antivirals effective in controlled clinical trials had unbound Cmax/EC50 ≥ 6.8 in Calu-3 or Caco-2 cells. EC50 of nucleoside analogs were cell dependent. This approach and earlier availability of more relevant cultures could have reduced the number of unwarranted clinical trials.
Full article
(This article belongs to the Special Issue Advances in Antiviral Agents against SARS-CoV-2 and Its Variants 2nd Edition)
►▼
Show Figures

Graphical abstract
Open AccessCommunication
Association of the IFNG +874T/A Polymorphism with Symptomatic COVID-19 Susceptibility
by
Kevin Matheus Lima de Sarges, Flávia Póvoa da Costa, Erika Ferreira dos Santos, Marcos Henrique Damasceno Cantanhede, Rosilene da Silva, Adriana de Oliveira Lameira Veríssimo, Maria de Nazaré do Socorro de Almeida Viana, Fabíola Brasil Barbosa Rodrigues, Mauro de Meira Leite, Maria Karoliny da Silva Torres, Christiane Bentes da Silva, Mioni Thieli Figueiredo Magalhães de Brito, Andréa Luciana Soares da Silva, Daniele Freitas Henriques, Izaura Maria Vieira Cayres Vallinoto, Giselle Maria Rachid Viana, Maria Alice Freitas Queiroz, Antonio Carlos Rosário Vallinoto and Eduardo José Melo dos Santos
Viruses 2024, 16(4), 650; https://doi.org/10.3390/v16040650 - 22 Apr 2024
Abstract
Tumor necrosis factor (TNF) and interferon-gamma (IFNγ) are important inflammatory mediators in the development of cytokine storm syndrome (CSS). Single nucleotide polymorphisms (SNPs) regulate the expression of these cytokines, making host genetics a key factor in the prognosis of COVID-19. In this study,
[...] Read more.
Tumor necrosis factor (TNF) and interferon-gamma (IFNγ) are important inflammatory mediators in the development of cytokine storm syndrome (CSS). Single nucleotide polymorphisms (SNPs) regulate the expression of these cytokines, making host genetics a key factor in the prognosis of COVID-19. In this study, we investigated the associations of the TNF -308G/A and IFNG +874T/A polymorphisms with COVID-19. We analyzed the frequencies of the two polymorphisms in the control groups (CG: TNF -308G/A, n = 497; IFNG +874T/A, n = 397), a group of patients with COVID-19 (CoV, n = 222) and among the subgroups of patients with nonsevere (n = 150) and severe (n = 72) COVID-19. We found no significant difference between the genotypic and allelic frequencies of TNF -308G/A in the groups analyzed; however, both the frequencies of the high expression genotype (TT) (CoV: 13.51% vs. CG: 6.30%; p = 0.003) and the *T allele (CoV: 33.56% vs. CG: 24. 81%; p = 0.001) of the IFNG +874T/A polymorphism were higher in the COVID-19 group than in the control group, with no differences between the subgroups of patients with nonsevere and severe COVID-19. The *T allele of IFNG +874T/A (rs2430561) is associated with susceptibility to symptomatic COVID-19. These SNPs provided valuables clues about the potential mechanism involved in the susceptibility to developing symptomatic COVID-19.
Full article
(This article belongs to the Special Issue Basic Sciences for the Conquest of COVID-19)
Open AccessArticle
Characterizing Infections in Two Epidemic Waves of SARS-CoV-2 Omicron Variants: A Cohort Study in Guangzhou, China
by
Lin Qu, Chunyan Xie, Ming Qiu, Lina Yi, Zhe Liu, Lirong Zou, Pei Hu, Huimin Jiang, Huimin Lian, Mingda Yang, Haiyi Yang, Huiling Zeng, Huimin Chen, Jianguo Zhao, Jianpeng Xiao, Jianfeng He, Ying Yang, Liang Chen, Baisheng Li, Jiufeng Sun and Jing Luadd
Show full author list
remove
Hide full author list
Viruses 2024, 16(4), 649; https://doi.org/10.3390/v16040649 - 22 Apr 2024
Abstract
Background: After the adjustment of COVID-19 epidemic policy, mainland China experienced two consecutive waves of Omicron variants within a seven-month period. In Guangzhou city, as one of the most populous regions, the viral infection characteristics, molecular epidemiology, and the dynamic of population immunity
[...] Read more.
Background: After the adjustment of COVID-19 epidemic policy, mainland China experienced two consecutive waves of Omicron variants within a seven-month period. In Guangzhou city, as one of the most populous regions, the viral infection characteristics, molecular epidemiology, and the dynamic of population immunity are still elusive. Methods: We launched a prospective cohort study in the Guangdong Provincial CDC from December 2022 to July 2023. Fifty participants who received the same vaccination regimen and had no previous infection were recruited. Results: 90% of individuals were infected with Omicron BA.5* variants within three weeks in the first wave. Thirteen cases (28.26%) experienced infection with XBB.1* variants, occurring from 14 weeks to 21 weeks after the first wave. BA.5* infections exhibited higher viral loads in nasopharyngeal sites compared to oropharyngeal sites. Compared to BA.5* infections, the XBB.1* infections had significantly milder clinical symptoms, lower viral loads, and shorter durations of virus positivity. The infection with the BA.5* variant elicited varying levels of neutralizing antibodies against XBB.1* among different individuals, even with similar levels of BA.5* antibodies. The level of neutralizing antibodies specific to XBB.1* determined the risk of reinfection. Conclusions: The rapid large-scale infections of the Omicron variants have quickly established herd immunity among the population in mainland China. In the future of the COVID-19 epidemic, a lower infection rate but a longer duration can be expected. Given the large population size and ongoing diversified herd immunity, it remains crucial to closely monitor the molecular epidemiology of SARS-CoV-2 for the emergence of new variants of concern in this region. Additionally, the timely evaluation of the immune status across different age groups is essential for informing future vaccination strategies and intervention policies.
Full article
(This article belongs to the Special Issue COVID-19 Diagnostics in Clinical Applications and Pandemic Controls 2024)
►▼
Show Figures
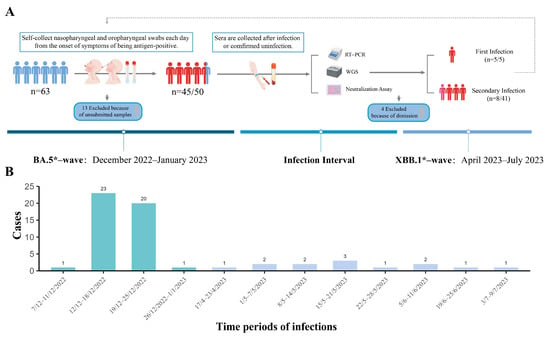
Figure 1
Open AccessArticle
Sosuga Virus Detected in Egyptian Rousette Bats (Rousettus aegyptiacus) in Sierra Leone
by
Brian R. Amman, Alusine H. Koroma, Amy J. Schuh, Immah Conteh, Tara K. Sealy, Ibrahim Foday, Jonathan Johnny, Ibrahim A. Bakarr, Shannon L. M. Whitmer, Emily A. Wright, Aiah A. Gbakima, James Graziano, Camilla Bangura, Emmanuel Kamanda, Augustus Osborne, Emmanuel Saidu, Jonathan A. Musa, Doris F. Bangura, Sammuel M. T. Williams, George M. Fefegula, Christian Sumaila, Juliet Jabaty, Fatmata H. James, Amara Jambai, Kate Garnett, Thomas F. Kamara, Jonathan S. Towner and Aiah Lebbieadd
Show full author list
remove
Hide full author list
Viruses 2024, 16(4), 648; https://doi.org/10.3390/v16040648 - 22 Apr 2024
Abstract
Sosuga virus (SOSV), a rare human pathogenic paramyxovirus, was first discovered in 2012 when a person became ill after working in South Sudan and Uganda. During an ecological investigation, several species of bats were sampled and tested for SOSV RNA and only one
[...] Read more.
Sosuga virus (SOSV), a rare human pathogenic paramyxovirus, was first discovered in 2012 when a person became ill after working in South Sudan and Uganda. During an ecological investigation, several species of bats were sampled and tested for SOSV RNA and only one species, the Egyptian rousette bat (ERBs; Rousettus aegyptiacus), tested positive. Since that time, multiple other species have been sampled and ERBs in Uganda have continued to be the only species of bat positive for SOSV infection. Subsequent studies of ERBs with SOSV demonstrated that ERBs are a competent host for SOSV and shed this infectious virus while exhibiting only minor infection-associated pathology. Following the 2014 Ebola outbreak in West Africa, surveillance efforts focused on discovering reservoirs for zoonotic pathogens resulted in the capture and testing of many bat species. Here, SOSV RNA was detected by qRT-PCR only in ERBs captured in the Moyamba District of Sierra Leone in the central region of the country. These findings represent a substantial range extension from East Africa to West Africa for SOSV, suggesting that this paramyxovirus may occur in ERB populations throughout its sub-Saharan African range.
Full article
(This article belongs to the Special Issue Bat- and Rodent-Borne Zoonotic Viruses)
►▼
Show Figures
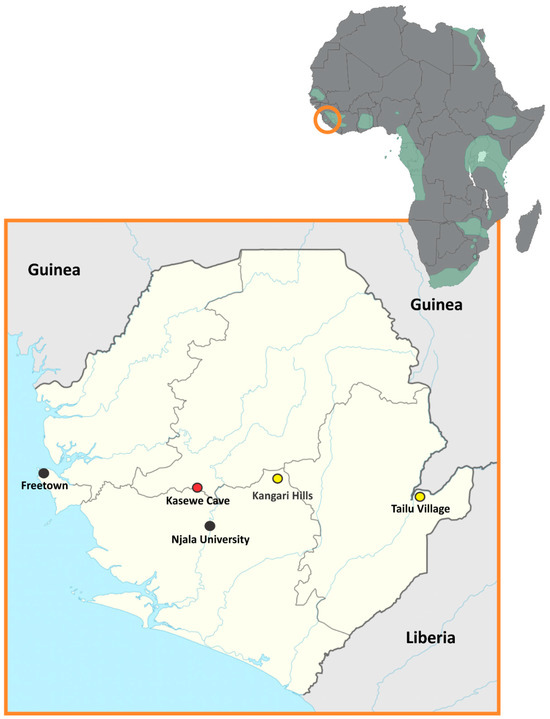
Figure 1
Open AccessReview
Back to the Basics of SARS-CoV-2 Biochemistry: Microvascular Occlusive Glycan Bindings Govern Its Morbidities and Inform Therapeutic Responses
by
David E. Scheim, Peter I. Parry, David J. Rabbolini, Colleen Aldous, Morimasa Yagisawa, Robert Clancy, Thomas J. Borody and Wendy E. Hoy
Viruses 2024, 16(4), 647; https://doi.org/10.3390/v16040647 - 22 Apr 2024
Abstract
Consistent with the biochemistry of coronaviruses as well established over decades, SARS-CoV-2 makes its initial attachment to host cells through the binding of its spike protein (SP) to sialylated glycans (containing the monosaccharide sialic acid) on the cell surface. The virus can then
[...] Read more.
Consistent with the biochemistry of coronaviruses as well established over decades, SARS-CoV-2 makes its initial attachment to host cells through the binding of its spike protein (SP) to sialylated glycans (containing the monosaccharide sialic acid) on the cell surface. The virus can then slide over and enter via ACE2. SARS-CoV-2 SP attaches particularly tightly to the trillions of red blood cells (RBCs), platelets and endothelial cells in the human body, each cell very densely coated with sialic acid surface molecules but having no ACE2 or minimal ACE2. These interlaced attachments trigger the blood cell aggregation, microvascular occlusion and vascular damage that underlie the hypoxia, blood clotting and related morbidities of severe COVID-19. Notably, the two human betacoronaviruses that express a sialic acid-cleaving enzyme are benign, while the other three—SARS, SARS-CoV-2 and MERS—are virulent. RBC aggregation experimentally induced in several animal species using an injected polysaccharide caused most of the same morbidities of severe COVID-19. This glycan biochemistry is key to disentangling controversies that have arisen over the efficacy of certain generic COVID-19 treatment agents and the safety of SP-based COVID-19 vaccines. More broadly, disregard for the active physiological role of RBCs yields unreliable or erroneous reporting of pharmacokinetic parameters as routinely obtained for most drugs and other bioactive agents using detection in plasma, with whole-blood levels being up to 30-fold higher. Appreciation of the active role of RBCs can elucidate the microvascular underpinnings of other health conditions, including cardiovascular disease, and therapeutic opportunities to address them.
Full article
(This article belongs to the Special Issue Glycans in Virus-Host Interactions)
►▼
Show Figures
Figure 1

Journal Menu
► ▼ Journal Menu-
- Viruses Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Diseases, Infectious Disease Reports, Pathogens, Viruses, TropicalMed
Human Monkeypox Research
Topic Editors: Shailendra K. Saxena, Ahmed Sayed Abdel-MoneimDeadline: 30 June 2024
Topic in
Biomedicines, JCM, Pathogens, Vaccines, Viruses
Discovery and Development of Monkeypox Disease Treatments
Topic Editors: Mohd Imran, Ali A. RabaanDeadline: 31 August 2024
Topic in
Brain Sciences, Clinics and Practice, COVID, Life, Vaccines, Viruses
Multifaceted Efforts from Basic Research to Clinical Practice in Controlling COVID-19 Disease
Topic Editors: Yih-Horng Shiao, Rashi OjhaDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
Viruses
Oncolytic Viruses as Immunotherapeutic Agents
Guest Editor: Nadine Van MontfoortDeadline: 1 May 2024
Special Issue in
Viruses
Animal Coronaviruses: Infection, Prevention, and Antivirals
Guest Editor: Tomomi TakanoDeadline: 20 May 2024
Special Issue in
Viruses
Bacteriophages and Biofilms 2.0
Guest Editors: Tomasz Olszak, Zuzanna Drulis-KawaDeadline: 31 May 2024
Special Issue in
Viruses
The Inflammasomes - Key Players in Antiviral Response
Guest Editors: Tsan Sam Xiao, Dong-Yan JinDeadline: 15 June 2024
Topical Collections
Topical Collection in
Viruses
Poxviruses
Collection Editors: Galileu Barbosa Costa, Giliane de Souza Trindade, Flavio Guimaraes da Fonseca
Topical Collection in
Viruses
Phage Therapy
Collection Editors: Nina Chanishvili, Jean-Paul Pirnay, Mikael Skurnik
Topical Collection in
Viruses
Coronaviruses
Collection Editors: Fernando Almazan Toral, Luis Martinez-Sobrido
Topical Collection in
Viruses
SARS-CoV-2 and COVID-19
Collection Editors: Fernando Almazan Toral, Luis Martinez-Sobrido








